 
“Omicron replicates very fast, so you may become very infectious within hours,” says Petersen. You could become infectious after you take the test, which is why scientists recommend taking it as close to your social plans as possible, ideally within an hour. How long before meeting family and friends should you take one? However, the gold standard remains the polymerase chain reaction (PCR) test, which detects viral load and infectiousness at lower levels, including before and after the time range in which they are picked up by LFTs. The main benefit of LFTs is they can be carried out quickly and easily at home, with a result in under 30 minutes, and that they don’t need to be sent to a lab. After carrying out tests, the UK Health Security Agency said LFTs have a “comparable sensitivity” for Omicron as for other Covid variants such as Delta. A recent paper demonstrated that LFTs, when used correctly, are likely to have a sensitivity above 80% and in many cases above 90%. The reliability of LFTs depends in part on whether you follow the instructions carefully, which is why studies give varying estimates of their efficacy, from 40% to 90%. “Please continue to test on a daily basis,” urges Irene Petersen, professor of epidemiology and health informatics at University College London. If you have come into contact with an infected person, it could take a few days for a positive result to show up. LFTs identify specific viral proteins that are present inside your nose when coronavirus is infecting you. "Once the package is at room temperature, you may open it and perform the test according to the authorized instructions for use," the FDA says.How do LFTs work and how reliable are they, including for Omicron? The FDA recommends that people should leave tests unopened at room temperature for about two hours before being used if they have been delivered in freezing conditions or exposed to heat. It's also important to store the at-home tests properly, at room temperature in a dry location, he says. "You may get false negatives" even though you are actually infected. If the test is past its shelf life, "you're not going to get accurate results," Amiji says. "You're testing to see if the antibody will react to the antigen and produce a color product," he says. If the mucus contains the protein antigen of the SARS-CoV-2 virus, that means infection is present. 
The federal agency also suggests that people consider testing before coming into contact with someone at high risk for severe COVID-19, older adults and people who are immunocompromised, especially if they reside in an area with a medium to high COVID-19 hospital admission level.Īmiji says the at-home test kits work by putting antibodies in the test kit solution into contact with material from nasal swabs. Those who have been exposed to an infected person but do not have symptoms should wait a full five days after exposure to test themselves. The FDA says people with possible COVID-19 symptoms should test immediately. If it says to check the expiration date on the box and that date has passed, throw the test away. The FDA provides a link to products with extended expiration dates. People can also go directly to the FDA's website on expiration dates for a more extensive list of test brands. Some lots associated with InteliSwab, for instance, had expiration dates extended from October 2022 to January 2024. The lot number is next to the expiration date and may be a combination of letters and numbers. The North Carolina Department of Health and Human Services has a user-friendly website where people can use the test brand name and lot number to see if the FDA has revised the expiration date, Amiji says. He says there are a couple of ways to check to see whether the test kit you have is still effective. In some cases, the FDA approved extending the shelf life of tests by a year or so, Amiji says, adding that some tests retain their efficacy for 24 months after manufacture, with the new average being 20 months. 
How to find your test's expiration date online 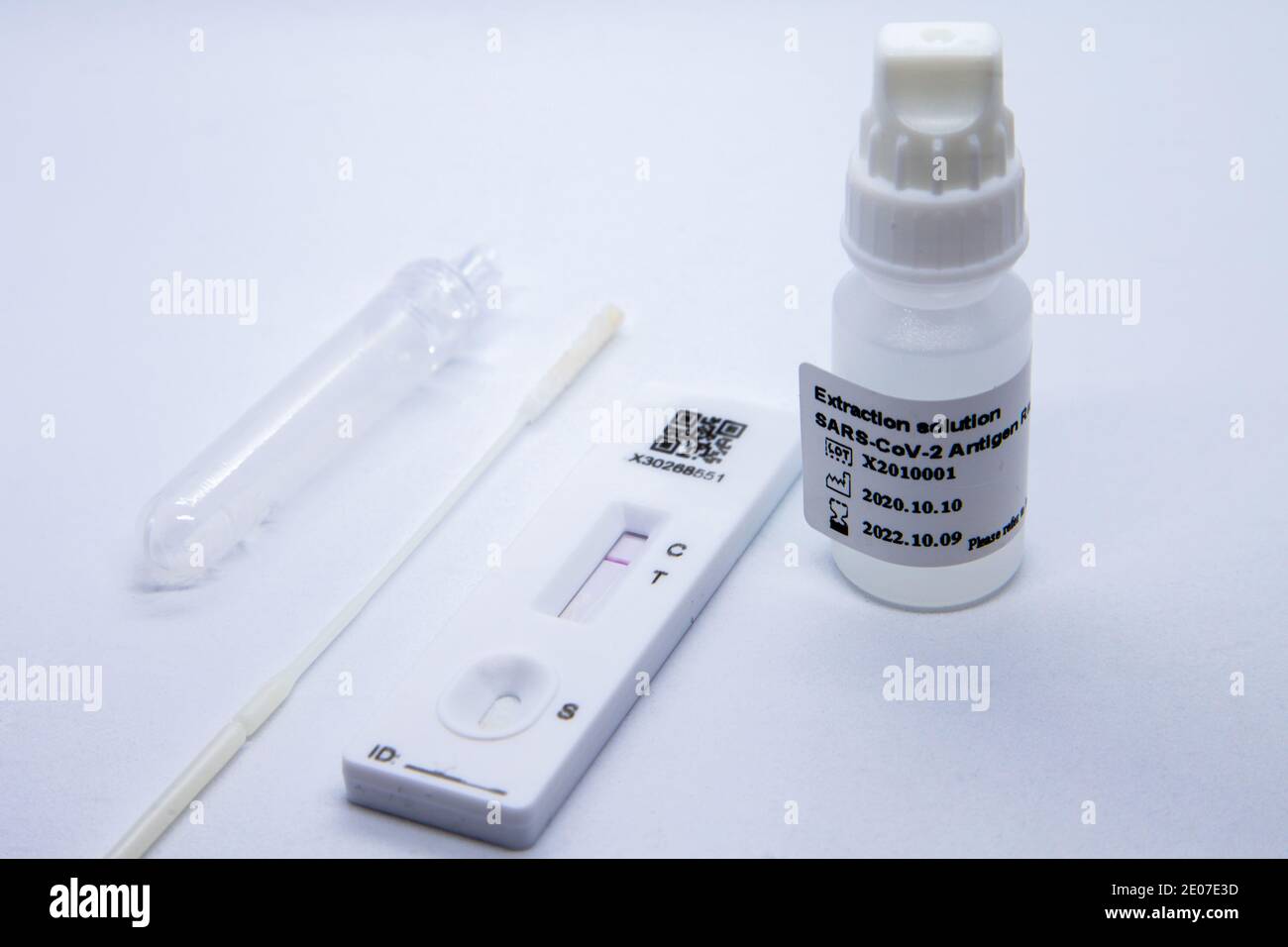
The federal agency initially said the tests expired after six to eight months of manufacture.īut since the first at-home antigen test for COVID-19 came out three years ago this month, test manufacturers have had time to conduct longer-range assessments of shelf stability. To be on the safe side, the FDA chose to be "very conservative" in terms of shelf life, he says. When the at-home tests for SARS-Cov-2 debuted, there was a lot of confusion about how reliable they would be for an extended period of time, Amiji says. He says the FDA has extended the shelf life of some common at-home antigen tests by months after new evaluations showed they remain stable for longer than first thought. Don't toss the test kit in the trash, says Mansoor Amiji, university distinguished professor of pharmaceutical sciences and chemical engineering. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
